
16 Mar How to enter the German Digital Health Market: Introduction to the DiGA
After a successful Digital Health delegation to Israel, GINSUM and MindUP hosted a webinar on the health landscape in Germany and its current developments in digital transformation. The partnership with the Israeli digital health incubator originated during the delegation trip in November 2021. These complex and delicate digitization processes offer a new set of challenges and opportunities for startups but also raise many questions. Our webinar, which took place on February 17, offered an introduction and insights into the complexities of the German health market and the best way to approach it.
In January 2021, the Federal cabinet in Germany passed the DVPMG act, a new law on digital modernization and the supply of care. Germany is the midst of a digital health revolution, aiming to improve the quality of health care and efficiency, and patients’ satisfaction, address long term challenges such as staff shortage and demographic change, and enable research and leverage potential for innovation. One of the main points of focus of the German Ministry of Health is focusing on the promotion of telemedicine and new market access for digital health applications- the DiGA (app on prescription). Implementation of the DiGA together with the development of e-prescriptions and research and data centers, are the key pillars for building a digital eco system in Germany. The DiGA’s have yet another important role, which is the tremendous impact they have in advancing research and innovation for future improvement of the care system, while the goal being the ability to export data into personal e-health records by 2023, explained Dr. Lars Hunze, Policy Advisor, German Federal Ministry of Health.
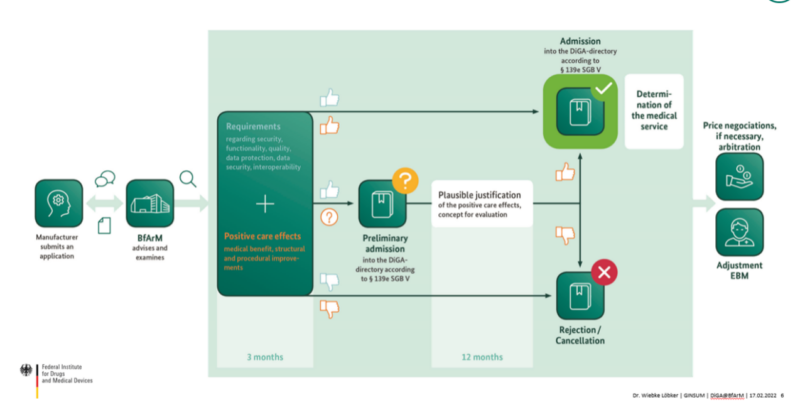
For a DiGA to be accepted into the system there is a specific process it must go through before it can be prescribed by physicians and or psychiatrist, and before it will be reimbursed by the statutory health insurance funds.
The approval of the DiGA’s is best made through the “fast track process of BfArM, the Federal Institute for Drugs and Medical Devices at the German Ministry of Health. BfArM is the department in charge, among other things, of the registration and evaluation of the risk factor in medical devices. In order to be listed in the DiGA directory, there are a few factors to comply with explained Dr. Wiebke Löbker, Innovation Office at BfArM. Factors that need to be taken into account are safety and performance (CE-marking needs to be according to MDD/MDR), general quality requirements, and the proof of positive health care effects. Proof can be shown either as a “medical benefit” (improvement of the state of health, extension of survival, shortening of the disease, etc.) and/or as an improvement of the healthcare structure and process related to the patient (coordination of treatment procedures, patient safety, reduction of the therapy-related efforts, etc).
The fast-track procedure’s assessment time is up to 3 months, during which BfArM offers consulting, guidance and support, to make the process as convenient as possible for the manufacturers and to ensure there is already sufficient evidence for a successful application process. Once BfArM receives an application it will decide if to support the app’s registration for a permanent admission or for a preliminary one. In case of being directed to the preliminary track, the manufacturer receives another year to try and prove the positive health care effects of the app, in order to get it enlisted in the permanent track. Once approved, price negotiations will be made separately between the manufacturers and the statutory health insurance. Also, once an app is certified, it can be found on the DiGA directory, where there are various search options for the end users convenience.
Currently up to date, BfArM received 122 applications, 8 applications are already under permanent listing, 22 are temporarily listed and need to hand in further proof, and 24 applications were rejected based on insufficient proof of positive health care effects.
As we can see there are various opportunities and windows for Israeli Digital Health companies, in the public health care sector as well as in the private one. In Germany we have 74 million statutory insurance users and 8.7 million private health insurance users. There are about 37 private insurance companies and it is important to understand that every German citizen is entitled to Statutory health insurance, but not everyone is entitled to purchase private insurance. In order to have access to private coverage a person needs a minimum income of 62-64 thousand Euros annually. Most people who are entitled to private insurance coverage are civilian service workers and state employed. So before entering the German market, it’s important to ask yourself if your product serves a special benefit to this specific type of population, suggests Estefania Rodriguez Migliarini, Start Ups Relations Manager, Insurtech Hub Munich. It is also interesting to note that, 72% of private insured customers said that they weren’t satisfied with the digital coverage. Furthermore, before trying to enter the German market, it is important to remember 3 key elements. First, sales cycles in Germany can be very long a weary. It can take up to 2 years and that is something that needs to be considered. Second, insurers are happy followers. If you obtain one good customers, others will be happy to follow, and third, Germans speak German!
Mr. Netanel Perez, Europe Regional Director, OrCam Technologies Ltd, a company focusing on assisting visually impaired people, further emphasized this point by explaining the importance of understanding the local mentality and work culture of the Germans. A key element to success is having a good connection and communication with the local stakeholders. Helpful would be having a local expert on the ground, acquainted with the specifications of the German market, while also receiving local feedback on the product and how it is perceived by the German customers. According to OrCam’s experience, once accepted into the system, the process works very smoothly. It’s also important to build your brand by building trust and awareness. This is done best by advertisement and participations at local exhibitions and conferences. Additional factors that can ease the process in Germany is to be included in the health catalogue of the GDP, and to have a local customer service with close relations to the distributors as well as to the end users and the insurance companies and to basically create your own personal eco-system. These steps are vital for constantly staying up to date. The more the market understands your product and its impact on society, the easier it will be to promote it.
OrCam is a great example since they were very successful in proving the success of their product. Thanks to their technology, Thousands of people at all levels, regained their independence through an improvement in their sight enabling them to return to work and integrate more easily in the day-to-day life.
Please feel free to contact the GINSUM team with any questions regarding the DiGA and the fast-track process and we will be happy to put you in touch with the relevant counter partner in Germany.
About the author

Jill Wyler
Jill Wyler held various professional positions in Israel and was until recently the Israel representative of a Washington based independent academic organization which focused on enhancing knowledge about modern Israel at universities in the United States and around the world.
At ELNET she works for the German Israeli Network of Startups & Mittelstand (GINSUM), as the Program Coordinator at the Israel office. Jill has a master’s degree in International Relations, with a focus on diplomacy and foreign policy, from the Hebrew University of Jerusalem

